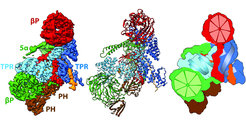
Structure of the human BBSome core complex
Klink BU, Gatsogiannis C, Hofnagel O, Wittinghofer A, Raunser S (2020). Elife
doi: 10.7554/eLife.53910.
Cilia are small, antenna-like structures on the surface of eukaryotic cells that harbor a unique set of sensory proteins, including GPCRs and other membrane proteins. The transport of these proteins requires the BBSome, recruited to the ciliary membranes by the G-Protein Arl6. How the BBSome core complex interacts with its ciliary cargo was already shown by the Raunser group and the Wittinghofer group in 2018.
In their latest publication they present a high-resolution cryo-EM structure of the human heterohexameric core subcomplex of the BBSome. The structure reveals the architecture of the complex in atomic detail. It explains how the subunits interact with each other and how disease-causing mutations hamper this interaction.