BUB1 and CENP-U, Primed by CDK1, Are the Main PLK1 Kinetochore Receptors in Mitosis
Singh P, Pesenti ME, Maffini S, Carmignani S, Hedtfeld M, Petrovic A, Srinivasamani A, Bange T, Musacchio A (2020) Mol Cell
Quelle
Reflecting its pleiotropic functions, Polo-like kinase 1 (PLK1) localizes to various sub-cellular structures during mitosis. At kinetochores, PLK1 contributes to microtubule attachments and mitotic checkpoint signaling. Previous studies identified a wealth of potential PLK1 receptors at kinetochores, as well as requirements for various mitotic kinases, including BUB1, Aurora B, and PLK1 itself. Here, we combine ectopic localization, in vitro reconstitution, and kinetochore localization studies to demonstrate that most and likely all of the PLK1 is recruited through BUB1 in the outer kinetochore and centromeric protein U (CENP-U) in the inner kinetochore. BUB1 and CENP-U share a constellation of sequence motifs consisting of a putative PP2A-docking motif and two neighboring PLK1-docking sites, which, contingent on priming phosphorylation by cyclin-dependent kinase 1 and PLK1 itself, bind PLK1 and promote its dimerization. Our results rationalize previous observations and describe a unifying mechanism for recruitment of PLK1 to human kinetochores.
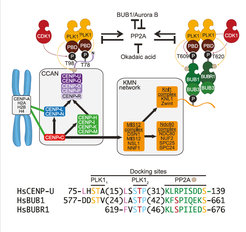
Up: Scheme of main findings on the mechanism of kinetochore recruitment of PLK1.Down: Sequence alignment of various mitotic and post-anaphase receptors of PLK1. PLK1 substrate sites or motifs are in yellow; CDK target sites in red within a blue motif; a hydrophobic residue enhancing target binding and dimerization is in magenta; PP2AB56 docking motif is in green. Numbers in parentheses indicate residues in sequence gaps.
