C-H Bond Activation for the Synthesis of Heterocyclic Atropisomers Yields Hedgehog Pathway Inhibitors
Shan G, Flegel J, Li H, Merten C, Ziegler S, Antonchick AP, Waldmann H (2018). Angew Chem Ind Ed Engl
doi: 10.1002/anie.201809680.
Described here is a rhodium (III)-catalyzed C@H bond activation and annulation for the atroposelective synthesis of axially chiral 4- arylisoquinolones. The method employs chiral cyclopentadienyl ligands embodying a piperidine ring as backbone and yields the atropisomers with good to excellent yields and enantioselectivity. Biological relevance of the 4-arylisoquinolones was demonstrated by their investigation in different cellular assays, leading to the discovery of novel non-SMO (SMO= smoothened) binding Hedgehog pathway inhibitors.
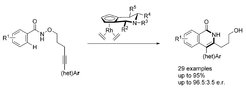
Atroposelective synthesis of 4-arylisoquinolones by RhIII-catalyzed C-H bond activation and intramolecular annulation.
