Enantioselective Formal C(sp3 )-H Bond Activation in the Synthesis of Bioactive Spiropyrazolone Derivatives.
Li H, Gontla R, Flegel J, Merten C, Ziegler S, Antonchick AP, Waldmann H (2018). Angew Chem Int Ed Engl doi: 10.1002/anie.201811041.
The Waldmann lab together with the Antonchick lab report the first enantioselective anulation of a-arylidene pyrazolones through a formal C(sp3)-H activation under mild conditions enabled by highly variable Rhodium(III)-Cpx catalysts.
The method gave access to a set of structurally diverse spiropyrazolones containing all-carbon quaternary centers in high yields and with high enantioselectivity. Preliminary biological investigations in different cellular assays led to the identification of the spiropyrazolones as a novel class of Hedgehog pathway inhibitors.
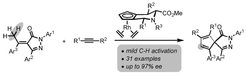
Enantioselective C-H activation/spiroannulation reactions using RhIII-Cpx catalysts
