How peroxisomes catch their enzymes
Experts from different fields together reveal the first structure of the peroxisomal docking complex
The cell contains numerous vesicles of different sizes. These so-called organelles are reaction spaces enclosed by a membrane, in which certain tasks are performed by enzymes: While energy is produced in the mitochondria, the power plants of the cell, dozens of enzymes work in the peroxisomes on the synthesis of lipids and on the detoxification of the cell by degradation of harmful fatty acids. The harmful toxic hydrogen peroxide produced during this process is neutralized in the peroxisomes. Defects of the peroxisomes or their enzymes lead to severe diseases with mostly fatal consequences.
Signal sequences guide enzymes to the right target
Most organelles, like peroxisomes, cannot produce their own enzymes. These are produced at the ribosomes and must then be correctly distributed to the respective organelles. For this purpose, the peroxisomal enzymes carry one of two specific signal sequences that are recognized by import receptors in the cytosol. Ralf Erdmann's group has already been able to show in earlier work that there is an extra pore for each of the two signal sequences, which transports the loaded receptor into the interior of the peroxisomes. "So far, however, only a few details about this process are known," Erdmann says, who has been studying peroxisomes for more than 30 years.
A central role is assigned to a docking complex that directs the loaded receptors to the peroxisomal membrane and traps them there. How this complex is formed by the proteins Pex13p, Pex14p and Pex17p and how it functions in detail still remains mysterious. Now, using electron microscopy, the team of Christos Gatsogiannis has succeeded for the first time in determining the architecture of the complex and thus gaining new mechanistic insights.
"The close cooperation of experts from different fields was a key to the success of this project," Bettina Warscheid says.
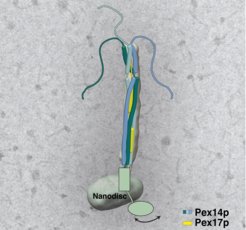
Fishing for enzymes
Tobias Hansen succeeded in isolating a highly pure complex of the proteins Pex14p and Pex17p as part of his doctoral thesis at the University of Bochum. Using native mass spectrometry, Daniel Wendscheck, a doctoral student at the University of Freiburg, was able to determine that the complex is composed of a total of three copies of the Pex14p protein and one Pex17. Pascal Lill, PhD student at the MPI in Dortmund reconstituted the docking complex in a nearly native membrane-like environment (Fig. 1) to simulate the conditions at the peroxisomal membrane. By applying cryo-electron microscopy and tomography he obtained the structure of the complex. It shows elongated strands arranged parallel to each other, which together form an overall rod-like structure. About two-thirds of this "rod" protrudes from the peroxisomes. Through further experiments, the three groups were finally able to clarify the exact arrangement of the proteins in an exciting process. At the end of the structure, the three Pex14p proteins show flexible areas that protrude into the space of the cell like tentacles (Fig. 2). The scientists suspect that the tentacles serve to fish for as many loaded receptors as possible.

"This complex is essential for functional peroxisomes. Our first picture of its structure and a mechanistic model of its function form an important basis for further studies of peroxisomes and could also contribute to a better understanding of peroxisomal diseases," Christos Gatsogiannis explains.
The protagonists of the project
Bettina Warscheid is head of the Department of Biochemistry and Functional Proteomics at the Institute of Biology II at the University of Freiburg. Ralf Erdmann is Head of the Department of Systems Biochemistry at the Institute of Biochemistry and Pathobiochemistry at the University of Bochum. Christos Gatsogiannis was a project goup leader at the Max-Planck-Institute of Molecular Physiology in Dortmund until recently. He accepted an appointment as Professor of Electron Tomography of Nanostructured Systems at the Institute of Medical Physics and Biophysics at the Medical Faculty of the University of Münster in September and will soon establish his group at the new "Center for Nanoscience. The collaboration took place within the framework of the research group "Structure and Function of the Peroxisomal Translocon" funded by the German Research Foundation.

