
Our research techniques
We benefit from a wide range of scientific techniques. These methods and the associated expertise are available to our scientists and researchers.

Cell Screening
Biochemical and cell-based screens are carried out using a state-of-the-art robotic system. The results are combined centrally and analyzed chemically using computer technology. The robotic system has a F5 robotic arm, a liquid handling station Freedom EVO100, an ECHO acoustic dispenser, a multidrop bulk dispenser and a paradigm multi-mode microplate reader.
COMAS >

Fluorescence Microscopy
To investigate the localization, interaction and activity of proteins in the cell, they are firstly labelled with fluorescent proteins. These proteins absorb light of a certain wavelength and emit light of a longer wavelength. The following widefield and confocal microscopes are available for fluorescence microscopy: Leica DM IRB, Motic AE31, Olympus Cell^R, Olympus Anisotropy, SAM, TIRF, LoTTi, Leica SP 5, Leica SP 8, Nikon, Olympus FV1000, Zeiss LSM 510.

Cryo-Electron Microscopy
Cryo-electron microscopy is used to visualize the structure of large protein complexes. Laborious crystallization of the proteins is not required. By means of fast-freezing technology, the protein complexes are conserved under nearly physiological conditions and the examined on a cooling table in the transmission electron microscope. For the analysis of cryo samples and chemically fixed specimens we have a 120kV transmission electron microscope (TEM) JEOL JEM 1400, a 300kV cryoTEM JEOL JEM 3200FSC and a 300 kV Titan Krios (FEI).

X-ray Crystallography
X-ray crystallography provides high-resolution 3D structures of proteins. We are equipped with a Rigaku X-ray generator with two MAR345 desktop beamlines, one of which has an automatic sample changer. This allows efficient in-house testing of small crystals. Protein samples of approximately 50 nL in size can be tested with a mosquito crystallization robot. We also have a Zinsser Lissy robotic system for pipetting master blocks and custom screens.

Mass Spectometry
Mass spectrometry is used to characterize chemical compounds, to analyze peptides, in the analysis of protein modifications and in proteome research. For this purpose, we have GC/EI-MS, HPLC/ESI-MS, GC/EI-HRMS, GC/CI-HRMS, HPLC/ESI-MS, HPLC/APCI-MS, and MALDI-MS at our disposal. The analyses in proteomics are done qualitatively and/or as relative quantifications by means of SILAC, TMT-label or label free.
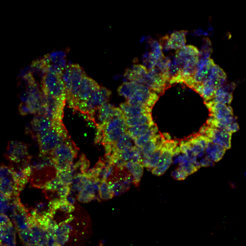
Organoid Culture
An organoid is a three-dimensional organ-bud grown in vitro that shows realistic micro-anatomy and thereby provides a realistic cellular model fot the investigation of human diseases. The technique for growing organoids has rapidly improved since the early 2010s, and it was named by “The Scientist“ as one of the biggest scientific advancements of 2013.





