Chemical Biology
The development of active substances that can specifically influence biological processes and possibly lead to improved treatment of diseases such as cancer is like looking for a needle in a haystack. Although millions, billions or even trillions of new molecules can be synthesised using synthesis robots, the probability of finding a useful substance among these molecules is very low.
We are therefore pursuing a different approach based on models found in nature. Evolution has yielded a wide variety of highly effective biologically active substances. Whether plants, animals, bacteria or fungi, each has substances that fulfil important tasks in the organism – usually by binding to a specific protein receptor. As has often been shown, some of these substances not only affect their actual target, but also influence human cells. The plant substances morphine and digitalis, from which highly effective medications for pain or heart disease have emerged, are well known examples of this.
Our research focuses on the development of methods and strategies for the production of a whole collection of active substances that are closely related to individual natural products. For this, we not only use numerous techniques of organic chemistry, but also biochemical and cell-based analysis assays and state-of-the-art methods of computer science. An ingenious computer programme called Scaffold Hunter, with which we search for promising scaffolds for the synthesis of suitable chemical tools for our experiments, was developed in our institute.
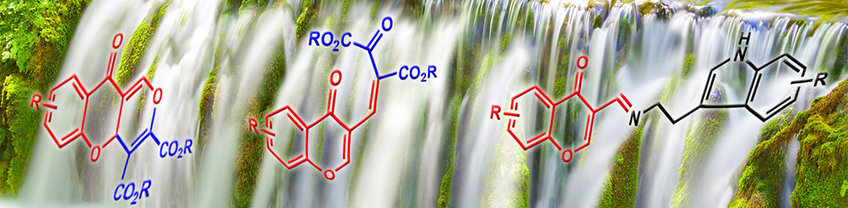
The cascade synthesis of centrocountins, a novel reaction cascade we developed, is a promising starting point for the development of innovative tumour therapies. Cancer cells treated with centrocountins do not divide into two, but rather into three or more daughter cells that are then no longer viable. Another focus in our department is the synthesis and study of lipidated proteins. They play an important role in signalling and transport processes in living cells. Our goal here is to develop highly practical and reliable methods for the synthesis of such lipoproteins, in particular those that belong to the group of Ras family proteins. We are seeking to identify small molecules to regulate the function and activity of this important protein family. Ras is a key component of various signal transduction pathways that regulate growth and differentiation processes – and is thus one of the central starting points for new, hopefully more effective cancer treatments.
Cells exposed to centrocountines (right) do not divide into two daughter cells as untreated cells (left), but rather into three daughter cells.